Melting Point Determination by DSC: Accurate and Reliable Methods
Melting Point Determination by DSC: Accurate and Reliable Methods
Melting Point Determination by DSC: Accurate and Reliable Methods
Why DSC Is the Gold Standard for Melting Point Analysis
While several methods exist for measuring melting points, DSC has become the gold standard for accurate, reproducible melting point determination across all material types. Unlike visual melting point apparatus that relies on subjective observation, DSC provides objective, quantitative data including both the melting temperature and the associated enthalpy change.
DSC offers several advantages over traditional methods: it requires only a few milligrams of sample, provides a precise onset temperature that is less sensitive to heating rate than visual methods, simultaneously measures the heat of fusion, and generates a permanent digital record of the measurement.
Pharmaceutical, chemical, polymer, and materials science industries all rely on DSC for definitive melting point data that is accepted worldwide for regulatory submissions, quality certificates, and specification compliance.
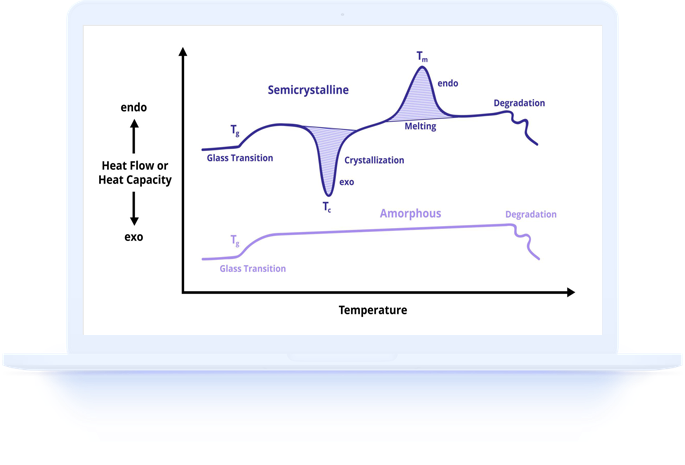
Understanding Melting Peaks in DSC Curves
The melting peak in a DSC curve contains rich information beyond just the melting temperature. The peak shape, width, and enthalpy all provide insights into the crystalline structure, purity, and thermal history of the sample.
A sharp, narrow melting peak indicates a highly crystalline, pure material with a well-defined crystal structure. A broad melting peak may indicate a distribution of crystal sizes, the presence of impurities, or a mixture of different crystalline forms (polymorphs). Multiple melting peaks suggest polymorphism, blend composition, or melting-recrystallization-remelting phenomena.
The melting endotherm area, measured in joules per gram, represents the heat of fusion and is proportional to the amount of crystalline material present. This quantitative information distinguishes DSC from simpler melting point methods that only provide a temperature value.
Onset vs Peak Temperature: Which to Report
Two temperatures are commonly reported from a DSC melting peak: the extrapolated onset temperature and the peak temperature. Understanding the difference is important for accurate communication and comparison of results.
The onset temperature is determined by the intersection of the baseline with the tangent to the leading edge of the melting peak. It represents the temperature at which melting first begins and is less sensitive to heating rate, sample mass, and thermal lag than the peak temperature. Most ASTM and ISO standards recommend reporting the onset temperature as the definitive melting point.
The peak temperature occurs at the maximum deviation of heat flow from the baseline and is always higher than the onset temperature. The difference between onset and peak temperatures increases with faster heating rates and larger sample masses. Peak temperature is sometimes reported in addition to onset temperature, particularly in pharmaceutical applications where it may have clinical relevance.
Factors Affecting Melting Point Measurements
Several factors affect the accuracy of melting point measurements by DSC. Heating rate is the most significant: faster rates produce higher apparent melting temperatures due to thermal lag between the sensor and the interior of the sample. Standard methods specify 10 degrees per minute, but rates of 1 to 5 degrees per minute give more accurate absolute values.
Sample mass affects peak shape and apparent melting temperature. Larger samples produce broader peaks and more thermal lag, shifting the peak temperature upward. Keeping sample mass between 2 and 5 milligrams minimizes these effects while maintaining adequate signal.
Particle size can affect the melting onset when surface-to-volume ratio changes the heat transfer efficiency. Grinding samples to a uniform fine powder before DSC analysis improves reproducibility, particularly for pharmaceutical and chemical purity determinations.
Sample-pan thermal contact is crucial: the sample should be distributed as a thin, flat layer on the bottom of the DSC pan. Poor contact causes erratic heat transfer and distorted peak shapes.
Melting Point of Polymers, Metals, and Pharmaceuticals
Different material classes show characteristic melting behavior in DSC that analysts must understand for proper interpretation. Polymers typically show broad melting ranges spanning 10 to 30 degrees Celsius, reflecting their lamellar crystal structure with a distribution of crystal thicknesses and perfection levels.
Small molecules, including drugs and pure chemicals, show sharp melting peaks that span only a few degrees when pure, broadening progressively with increasing impurity content. This sensitivity to impurities forms the basis of DSC purity analysis using the van’t Hoff method.
Metals and alloys show extremely sharp melting peaks due to their highly ordered crystal structures. Pure metals like indium, tin, and zinc melt at precisely defined temperatures and serve as calibration standards. Alloys show melting ranges that reflect their composition, with eutectic compositions showing sharp melting similar to pure metals.
Purity Assessment from Melting Point Data
The melting behavior of crystalline materials is sensitive to impurity content, a phenomenon described by the van’t Hoff equation. Impurities depress the melting onset temperature and broaden the melting peak, creating a characteristic shape that can be analyzed to determine purity.
The DSC purity method, described in ASTM E928, uses a mathematical analysis of the shape of the melting peak to calculate mole percent impurity without requiring knowledge of the identity of the impurities. Purities from about 95 to 99.99 mole percent can be determined, making this a valuable screening tool for pharmaceuticals and fine chemicals.
While the method has limitations and assumptions that restrict its applicability to certain types of samples, it remains one of the fastest and most convenient purity assessment techniques available, requiring only milligrams of sample and no reference standards or chromatographic separation.
Melting Point Testing Services
Our laboratory provides precise melting point determination by DSC for pharmaceuticals, polymers, chemicals, metals, and all other crystalline materials. We follow ASTM and ISO standard methods and report onset, peak, and endpoint temperatures along with heat of fusion data.
Contact us for a quotation on melting point testing services with standard or rush turnaround.

