DSC for Food Quality Testing: Fats, Proteins, and Starches
DSC for Food Quality Testing: Fats, Proteins, and Starches
DSC for Food Quality Testing: Fats, Proteins, and Starches
How DSC Is Used in the Food Industry
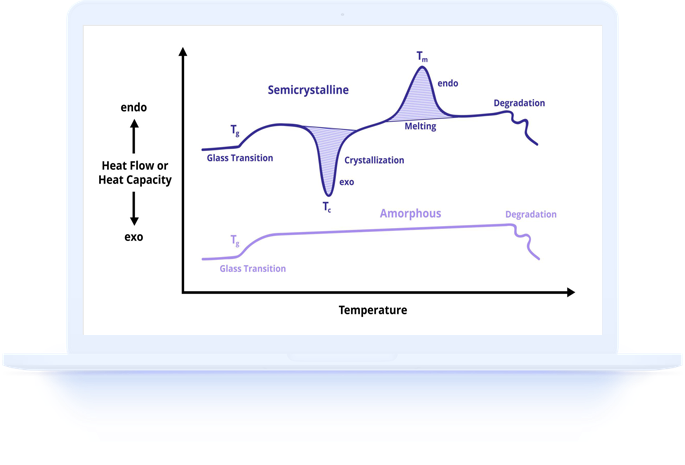
The food industry relies on thermal analysis to understand, control, and optimize the behavior of food materials during processing, storage, and consumption. DSC stands out as particularly valuable because it directly measures the energy changes associated with structural transformations in food components, providing fundamental data that relates to texture, stability, and sensory properties.
From farm to fork, thermal events shape food quality. Melting of fats determines chocolate texture and spread consistency. Protein denaturation governs the texture of cooked meats and baked goods. Starch gelatinization transforms raw flour into edible bread. Glass transitions in frozen and dried foods affect storage stability. DSC measures all of these phenomena quantitatively.
Modern food science increasingly uses DSC as part of formulation development programs, where thermal data helps predict how ingredient changes will affect product properties. This approach reduces the number of pilot-scale trials needed and accelerates the innovation cycle for new food products.
Fat and Oil Melting Profile Analysis
Fat melting behavior directly determines the sensory properties and functional performance of fat-based food products. DSC provides a complete melting profile showing the temperatures and enthalpies of all melting events, information that relates directly to product hardness, spreadability, and mouthfeel at different temperatures.
Different fats show characteristic DSC melting profiles that reflect their fatty acid composition and triglyceride structure. Butter shows a broad melting range from approximately minus 5 to 35 degrees Celsius, while coconut oil melts more sharply around 24 degrees Celsius. Palm oil exhibits a complex multi-peak profile reflecting its diverse triglyceride composition.
Solid fat content (SFC) at specific temperatures can be estimated from DSC melting curves by partial integration of the melting endotherm. This approach provides information similar to pulsed NMR measurements but from the same DSC scan used for other analyses. SFC profiles help formulators achieve desired product textures across different storage and usage temperatures.
Chocolate Tempering and Cocoa Butter Analysis
Chocolate quality depends critically on the crystalline form of cocoa butter, which can crystallize in six different polymorphic forms (labeled I through VI). Only Form V produces the desirable characteristics of glossy appearance, crisp snap, smooth mouthfeel, and resistance to bloom. DSC is the definitive tool for identifying cocoa butter polymorphs.
Each polymorphic form of cocoa butter has a characteristic melting point range: Form I melts at 17 degrees Celsius, Form II at 24 degrees, Form III at 26 degrees, Form IV at 28 degrees, the desirable Form V at 34 degrees, and Form VI at 36 degrees. DSC easily distinguishes these forms through their melting temperatures and enthalpies.
Tempering is the controlled crystallization process used to produce Form V crystals in chocolate. DSC provides an objective measure of tempering quality by analyzing the melting profile of finished chocolate. Properly tempered chocolate shows a single, sharp melting peak corresponding to Form V, while poorly tempered chocolate may show multiple peaks or a broader melting range.
Protein Denaturation and Stability Studies
Protein denaturation, the unfolding of the three-dimensional protein structure, is one of the most important thermal events in food processing. DSC measures the denaturation temperature (Td) and enthalpy of denaturation, which relate to protein stability and the conditions needed for cooking, pasteurization, and sterilization.
Each protein has a characteristic denaturation temperature that depends on its amino acid sequence and tertiary structure. Whey proteins denature around 70 to 80 degrees Celsius, while more heat-stable proteins like collagen denature at higher temperatures. The denaturation enthalpy reflects the extent of ordered structure in the native protein.
DSC protein analysis supports multiple food industry applications: optimizing pasteurization conditions to achieve microbial safety while minimizing protein damage, evaluating the effect of pH, salt concentration, and added ingredients on protein stability, and monitoring the degree of denaturation in processed food products as a quality indicator.
Starch Gelatinization and Retrogradation
Starch gelatinization, the irreversible transformation of native starch granules into a swollen, amorphous gel, is a fundamental event in the processing of bread, pasta, sauces, and countless other starch-based foods. DSC precisely measures the gelatinization temperature range and enthalpy, providing data essential for process optimization.
Different starch sources show characteristic gelatinization behavior. Corn starch gelatinizes over a range of approximately 65 to 75 degrees Celsius, while potato starch gelatinizes at slightly higher temperatures. Rice starch shows gelatinization behavior that varies with the amylose-to-amylopectin ratio, which differs between long-grain and short-grain varieties.
Starch retrogradation, the gradual recrystallization of gelatinized starch during storage, is responsible for bread staling and textural changes in many starch-based products. DSC measures retrogradation by detecting the small melting endotherm that develops in stored samples, providing quantitative data for evaluating anti-staling strategies and shelf-life studies.
Frozen Food Analysis by DSC
DSC plays an important role in the analysis of frozen foods, providing data about ice crystallization, glass transitions in the freeze-concentrated matrix, and the state of water within complex frozen systems. This information helps optimize freezing processes, predict storage stability, and maintain product quality.
When food is frozen, pure water forms ice crystals while dissolved solutes become concentrated in an unfrozen phase. At sufficiently low temperatures, this concentrated phase undergoes a glass transition (Tg prime) that determines the maximum storage stability temperature. DSC measures this critical temperature, typically found between minus 30 and minus 50 degrees Celsius for most food systems.
The amount of freezable water in a food system can be determined by DSC from the ice melting enthalpy. Bound water, tightly associated with proteins, starches, and other hydrophilic components, does not freeze and therefore does not contribute to the melting peak. The ratio of freezable to total water provides important information about water activity and microbial stability.
Food Quality Testing Services
Our food science DSC testing services help food manufacturers, ingredient suppliers, and research institutions characterize the thermal behavior of food materials for product development, quality control, and shelf-life assessment.
We offer comprehensive food DSC analysis including fat and oil melting profiles, chocolate tempering evaluation, protein denaturation studies, starch gelatinization and retrogradation measurements, frozen food glass transition analysis, and ingredient compatibility screening.
Our analysts are experienced in food-specific DSC applications and provide detailed interpretations relevant to your product development or quality control objectives. Contact us to discuss your food thermal analysis needs.

