DSC Enthalpy Measurement: Understanding Heat of Fusion and Reaction
DSC Enthalpy Measurement: Understanding Heat of Fusion and Reaction
DSC Enthalpy Measurement: Understanding Heat of Fusion and Reaction
What Is Enthalpy and Why It Matters
Enthalpy is a thermodynamic quantity that represents the total heat content of a system. In the context of DSC analysis, we are primarily interested in enthalpy changes that accompany physical transitions and chemical reactions in materials. These energy changes provide fundamental information about the nature and magnitude of thermal events.
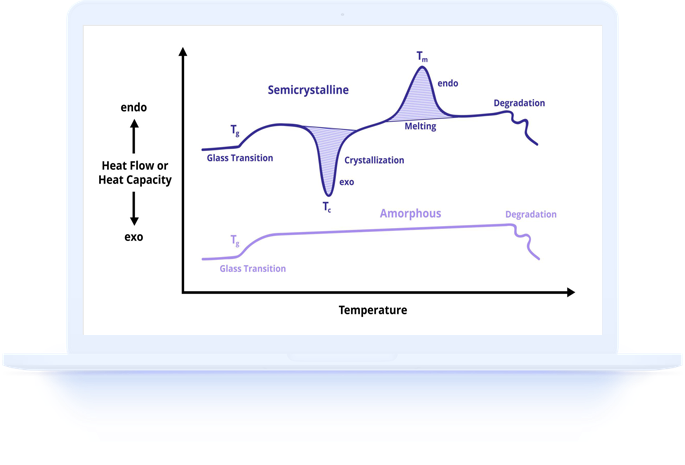
When a material melts, it absorbs a specific amount of energy called the heat of fusion or enthalpy of melting. When it crystallizes, it releases energy equal to the heat of crystallization. Chemical reactions such as curing, decomposition, and oxidation also involve characteristic enthalpy changes that DSC can precisely measure.
Understanding enthalpy values is essential because they provide quantitative data that goes beyond simply knowing at what temperature an event occurs. Two materials might melt at similar temperatures but have vastly different heats of fusion, indicating different molecular structures, crystalline forms, or degrees of crystalline perfection.
How DSC Measures Enthalpy Changes
DSC measures enthalpy changes by integrating the heat flow signal over time during a thermal event. The area enclosed between the DSC curve and the baseline during a peak represents the total energy absorbed or released during that event, expressed in millijoules or joules.
To convert the raw peak area to a specific enthalpy in joules per gram, the instrument divides the integrated area by the sample mass. Accurate sample weighing is therefore critical for reliable enthalpy measurements. Analytical balances with 0.01 milligram precision should be used, and the sample mass should typically be between 5 and 15 milligrams for optimal results.
The accuracy of enthalpy measurements depends on proper calibration, correct baseline selection, and appropriate integration limits. Enthalpy calibration using indium ensures that the instrument’s heat flow measurements are quantitatively correct. Regular verification of the calibration constant is essential for maintaining measurement quality.
Peak Area Integration Methods
Peak integration is the mathematical process of calculating the area under a DSC peak, and the choice of baseline construction method significantly affects the result. Modern DSC software offers several baseline types, and selecting the appropriate one requires understanding of the thermal event being measured.
A linear baseline, drawn as a straight line between the start and end of the peak, is suitable for well-resolved peaks on a flat baseline. A sigmoidal baseline, which follows an S-shaped curve, is preferred when there is a significant change in heat capacity across the transition, as commonly occurs during polymer melting.
For peaks that are partially overlapped with other thermal events, software deconvolution tools can mathematically separate the individual contributions. However, this approach introduces some uncertainty, and it is generally preferable to modify experimental conditions to achieve better peak resolution when possible.
Heat of Fusion: Melting and Crystallization Enthalpies
The heat of fusion (enthalpy of melting) is perhaps the most commonly measured enthalpy value in DSC. It represents the energy required to completely melt a crystalline material and provides direct information about the degree of crystalline order in the sample.
For pure, single-component crystalline materials, the heat of fusion is a characteristic physical property with a well-defined value. Indium has a heat of fusion of 28.45 joules per gram, water has 334 joules per gram, and ice provides one of the highest heats of fusion of common materials. These values reflect the strength of intermolecular forces holding the crystal lattice together.
In polymeric materials, the measured heat of fusion is less than the theoretical value for a fully crystalline polymer because real polymer samples always contain some amorphous material. The ratio of measured to theoretical heat of fusion gives the percent crystallinity, making this measurement essential for polymer characterization.
Reaction Enthalpies: Curing, Decomposition, and Oxidation
Chemical reactions produce characteristic enthalpy changes that DSC can measure to provide information about reaction completeness, kinetics, and energetics. Exothermic reactions such as polymerization, curing, and oxidation release heat, producing positive peaks in the DSC curve. Endothermic reactions such as dehydration and certain decomposition processes absorb heat, producing negative peaks.
The total enthalpy of a curing reaction indicates the total energy available from cross-linking and is used to calculate the degree of cure. A fully cured thermoset shows no residual exothermic peak upon reheating, while a partially cured sample shows a residual exotherm whose area corresponds to the remaining unreacted material.
Decomposition enthalpies measured by DSC provide information about the thermal stability and energy content of materials. This data is particularly important for safety assessments of energetic materials, pyrotechnics, and reactive chemical formulations where uncontrolled energy release could pose hazards.
Practical Applications of Enthalpy Data
Enthalpy data from DSC serves practical applications across numerous industries. In pharmaceutical quality control, the heat of fusion confirms the crystalline form and purity of drug substances, as different polymorphic forms have different enthalpies of melting.
In food science, enthalpy measurements of fat melting profiles determine the solid fat content at various temperatures, which directly relates to product texture and mouthfeel. Chocolate quality depends on the specific crystalline form of cocoa butter, identifiable by its characteristic melting enthalpy.
Materials scientists use enthalpy data for phase diagram construction, thermodynamic property databases, and computational modeling of material behavior. Accurate enthalpy values are essential inputs for process simulation software used in manufacturing and engineering design.
Energy storage researchers measure the enthalpy of phase-change materials (PCMs) to evaluate their capacity for thermal energy storage, a critical parameter for building insulation, thermal management systems, and portable heating and cooling devices.
Accurate Enthalpy Testing Services
Our laboratory provides precise enthalpy measurements using calibrated DSC instruments with documented accuracy and traceability. We measure heats of fusion, crystallization enthalpies, reaction enthalpies, and specific heat capacities for a wide range of materials and applications.
Our enthalpy testing services include standard melting and crystallization enthalpy measurements, degree of cure determination for thermoset materials, reaction enthalpy measurement for safety and process evaluation, and heat of solution and mixing studies. All measurements are performed following applicable ASTM or ISO standards.
Contact our technical team to discuss your specific enthalpy measurement needs and receive a detailed quotation.

