Crystallinity Measurement by DSC: Methods, Calculations, and Applications
Crystallinity Measurement by DSC: Methods, Calculations, and Applications
Crystallinity Measurement by DSC: Methods, Calculations, and Applications
Understanding Crystallinity in Materials
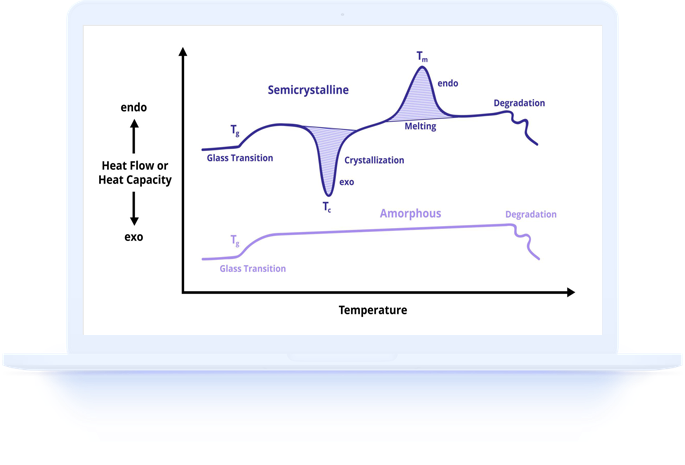
Crystallinity refers to the degree of structural order in a solid material. In polymers, chains can either arrange themselves in ordered, repeating patterns (crystalline regions) or remain in a disordered, tangled state (amorphous regions). Most semi-crystalline polymers contain both crystalline and amorphous regions, and the ratio between them profoundly affects material properties.
Higher crystallinity generally leads to greater stiffness, tensile strength, chemical resistance, and density, while lower crystallinity provides greater flexibility, transparency, and impact resistance. Understanding and controlling crystallinity is therefore essential for achieving desired product performance in applications ranging from packaging films to structural components.
The degree of crystallinity is influenced by the polymer’s chemical structure, molecular weight, processing conditions (cooling rate, orientation, annealing), and the presence of nucleating agents or plasticizers. DSC provides the most widely used method for quantifying crystallinity in polymeric materials.
How DSC Measures Crystallinity
DSC measures crystallinity by quantifying the enthalpy of melting, which is the energy required to break down the crystalline structure during heating. Since only the crystalline portion of the polymer contributes to the melting enthalpy, comparing the measured value to the theoretical value for a 100 percent crystalline sample gives the percent crystallinity.
A standard DSC measurement for crystallinity involves heating the sample through its melting range at a constant rate, typically 10 degrees Celsius per minute. The melting endotherm appears as a peak, and integrating the area under this peak gives the enthalpy of fusion in joules per gram. This measured enthalpy is then divided by the reference enthalpy for the fully crystalline form of that specific polymer.
For accurate crystallinity measurements, the baseline must be carefully selected for peak integration. Software tools provide various baseline construction options including linear, sigmoidal, and tangential baselines. The choice of baseline can significantly affect the calculated crystallinity, so using a consistent method is important for comparing samples.
Calculating Percent Crystallinity from DSC Data
The percent crystallinity calculation is straightforward: divide the measured heat of fusion by the heat of fusion for the 100 percent crystalline polymer, then multiply by 100. The formula is: Crystallinity (percent) = (measured enthalpy / reference enthalpy) times 100.
Reference enthalpy values for common polymers are well established in the scientific literature. Polyethylene has a reference value of 293 joules per gram, polypropylene is 207 joules per gram, PET (polyethylene terephthalate) is 140 joules per gram, and nylon 6 is 230 joules per gram.
When cold crystallization occurs during the heating scan (visible as an exothermic peak before the melting endotherm), its enthalpy should be subtracted from the melting enthalpy before calculating crystallinity. The corrected formula becomes: Crystallinity = (melting enthalpy minus cold crystallization enthalpy) divided by the reference enthalpy, times 100. This correction accounts for the fact that the cold crystallization formed during the DSC measurement rather than being present in the original sample.
Factors That Affect Crystallinity Measurements
Several experimental and material factors can affect crystallinity measurements by DSC. Heating rate influences the measured values because faster heating can cause superheating effects and incomplete melting of less stable crystalline forms. Standard methods typically specify 10 degrees per minute for consistency.
Sample thermal history significantly affects crystallinity. A rapidly cooled (quenched) sample will have lower crystallinity than a slowly cooled or annealed sample of the same polymer. For this reason, the first heating scan reflects the as-received crystallinity, while a second heating scan after controlled cooling reveals the inherent crystallization tendency of the material.
Multiple melting peaks, commonly observed in polymers like PET and nylon, can complicate crystallinity calculations. These multiple peaks may arise from different crystalline forms, lamellar thickness distributions, or melting-recrystallization-remelting sequences during the DSC scan. Proper peak integration that captures all melting events is essential for accurate total crystallinity values.
Cold Crystallization and Recrystallization in DSC
Cold crystallization is a phenomenon unique to semi-crystalline polymers that have been cooled too rapidly for complete crystallization. When these samples are subsequently heated in the DSC, the polymer chains gain enough mobility to crystallize during the measurement itself, producing an exothermic peak before the melting endotherm.
PET (polyethylene terephthalate) is the classic example of a polymer that readily shows cold crystallization. Rapidly cooled PET bottles and films can be nearly amorphous, showing a large cold crystallization exotherm during DSC heating around 120 to 140 degrees Celsius, followed by melting near 250 degrees Celsius.
The presence and magnitude of cold crystallization provides valuable information about processing conditions. A large cold crystallization peak indicates rapid cooling during fabrication, while its absence suggests slow cooling or deliberate annealing. Monitoring cold crystallization behavior helps processors optimize cooling conditions for desired transparency, barrier properties, and mechanical performance.
Industrial Applications of Crystallinity Testing
Crystallinity measurement by DSC serves critical functions across numerous industries. In polymer manufacturing, it provides a rapid quality control check for incoming resins and finished products, ensuring that processing conditions produced the target crystallinity level for optimal performance.
Packaging manufacturers monitor crystallinity in PET bottles, films, and containers because it directly affects clarity, barrier properties, and mechanical strength. Higher crystallinity improves barrier performance but reduces transparency, so precise control is essential.
Medical device manufacturers rely on crystallinity data for implantable devices made from ultra-high molecular weight polyethylene (UHMWPE), where crystallinity affects wear resistance and long-term performance. Regulatory submissions for medical devices commonly include DSC crystallinity data as evidence of proper processing.
Fiber and textile producers use DSC crystallinity measurements to optimize spinning and drawing conditions for synthetic fibers, where crystallinity and orientation together determine tenacity, modulus, and shrinkage behavior.
Expert Crystallinity Analysis Services
Our laboratory provides comprehensive crystallinity analysis services using state-of-the-art DSC instruments calibrated with traceable reference materials. We determine percent crystallinity for all common semi-crystalline polymers including polyethylene, polypropylene, PET, nylon, PEEK, and PVDF.
Our crystallinity testing services include standard first-heat crystallinity measurement reflecting as-received conditions, controlled crystallization studies to characterize the intrinsic crystallization behavior of your material, and comparative analyses for quality control, competitor benchmarking, or failure investigation.
All results are reported with detailed methodology documentation suitable for quality records, customer specifications, or regulatory submissions. Contact us to discuss your crystallinity testing requirements and receive a quotation.

